|
3/2/2024 0 Comments Molecular geometry table 7.4 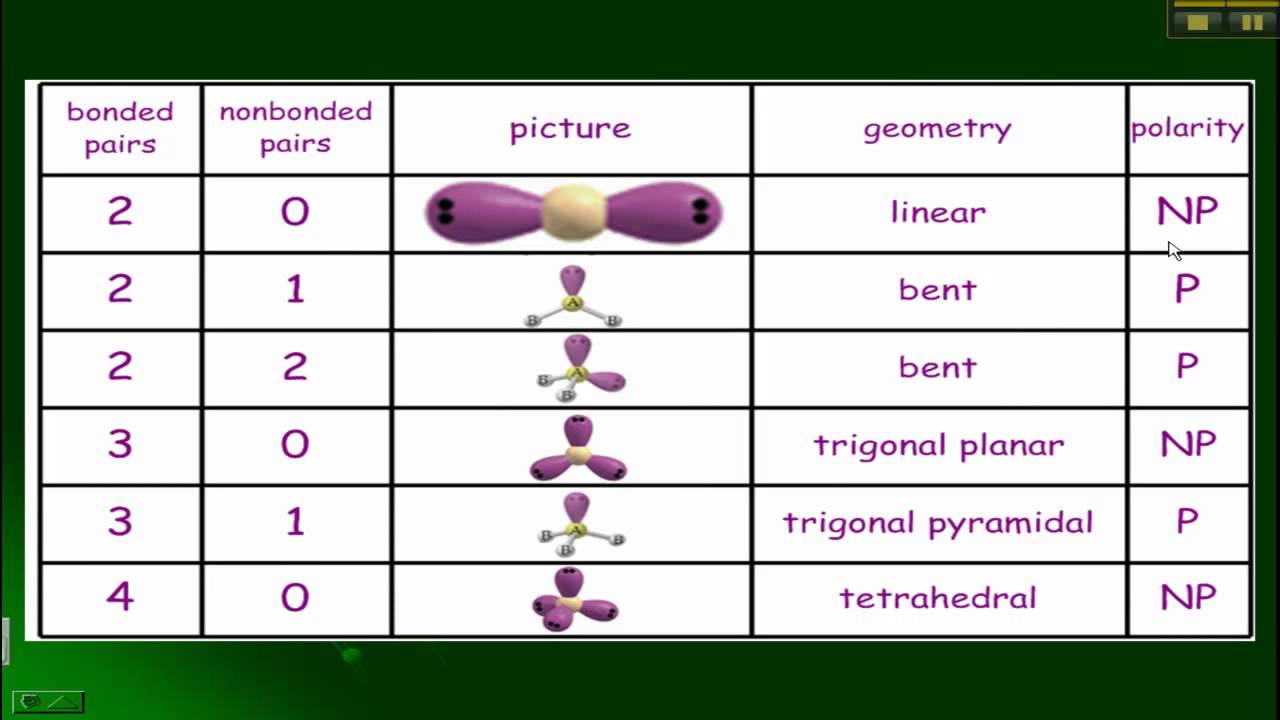
The electron-pair geometries shown in Figure 4.16 describe all regions where electrons are located, bonds as well as lone pairs. It is important to note that electron-pair geometry around a central atom is not the same thing as its molecular structure. 1: Molecular Orbitals for the H 2 Molecule. Electron-pair Geometry versus Molecular Structure. The hydronium ion, H 3 O +, forms when acids are dissolved in water. (b) Did you have to make any approximations or assumptions in completing your calculations (c) Is the concentration of citrate ion 1C6H5O7 3-2 equal to, less than, or greater than the. 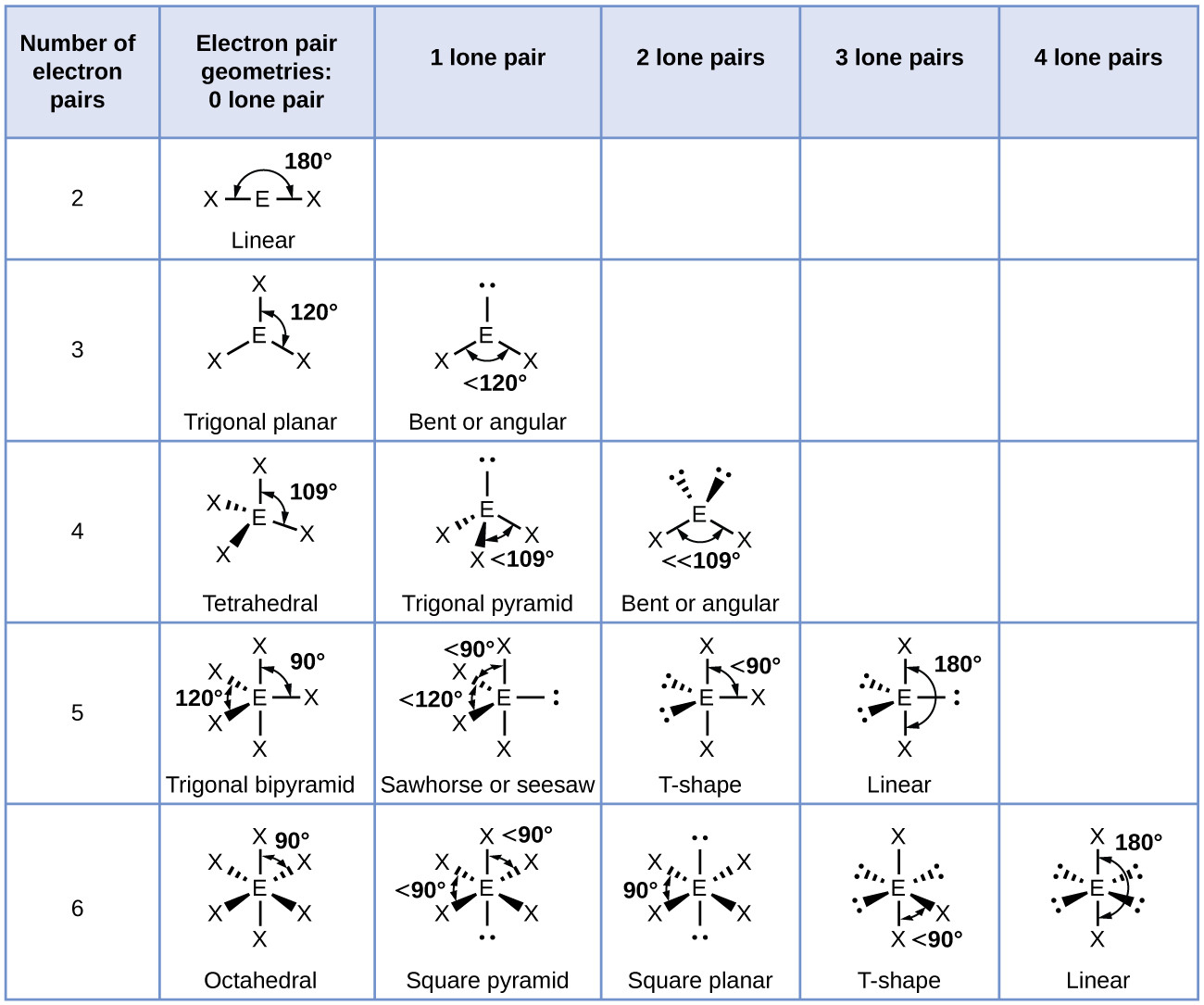
(a) Calculate the pH of a 0.040 M solution of citric acid. (b) Two of the electron regions are lone pairs, so the molecular structure is bent. Citric acid, which is present in citrus fruits, is a triprotic acid (Table 16.3). MOLECULAR SYMMETRY - Texas A&M University Understanding Character Tables. But it was not until 1985 that a new form of carbon was recognized: buckminsterfullerene, commonly known as a “buckyball.” Experimental evidence revealed the formula, C60, and then scientists determined how 60 carbon atoms could form one symmetric, stable molecule. A molecule must have as many molecular orbitals as there are atomic orbitals. 9: (a) H 2 O has four regions of electron density around the central atom, so it has a tetrahedral electron-pair geometry. NH2 - Molecular Geometry, Bond Angles (and Electron Geometry) Geometry of Molecules. 7.0: Prelude to Chemical Bonding and Molecular Geometry It has long been known that pure carbon occurs in different forms (allotropes) including graphite and diamonds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
